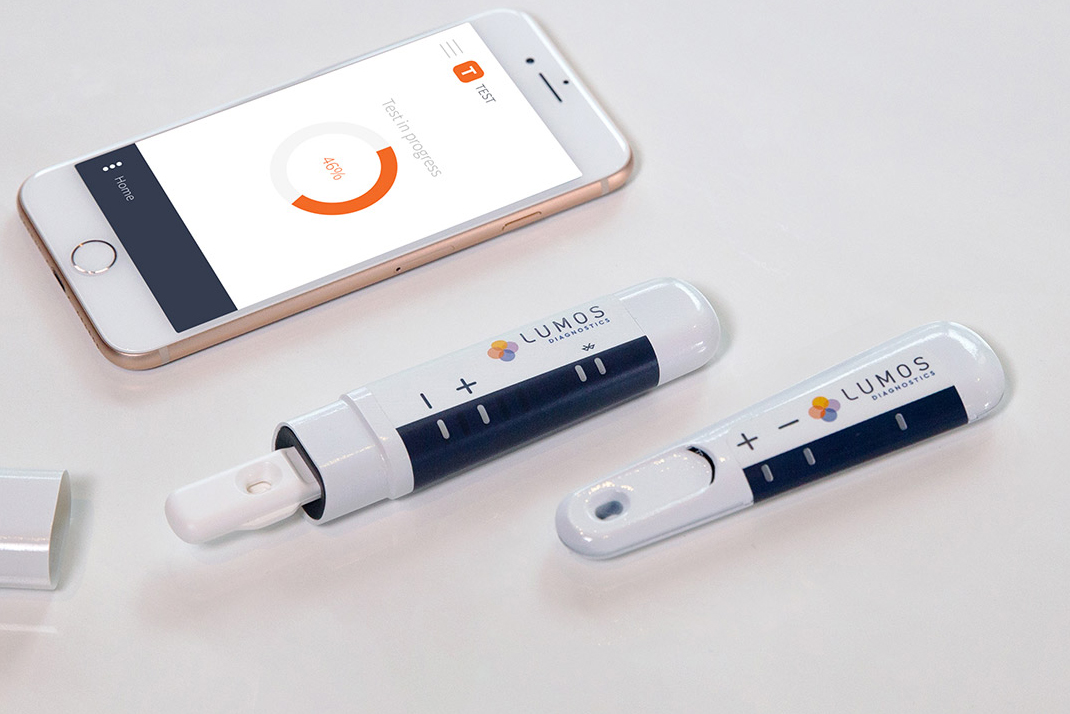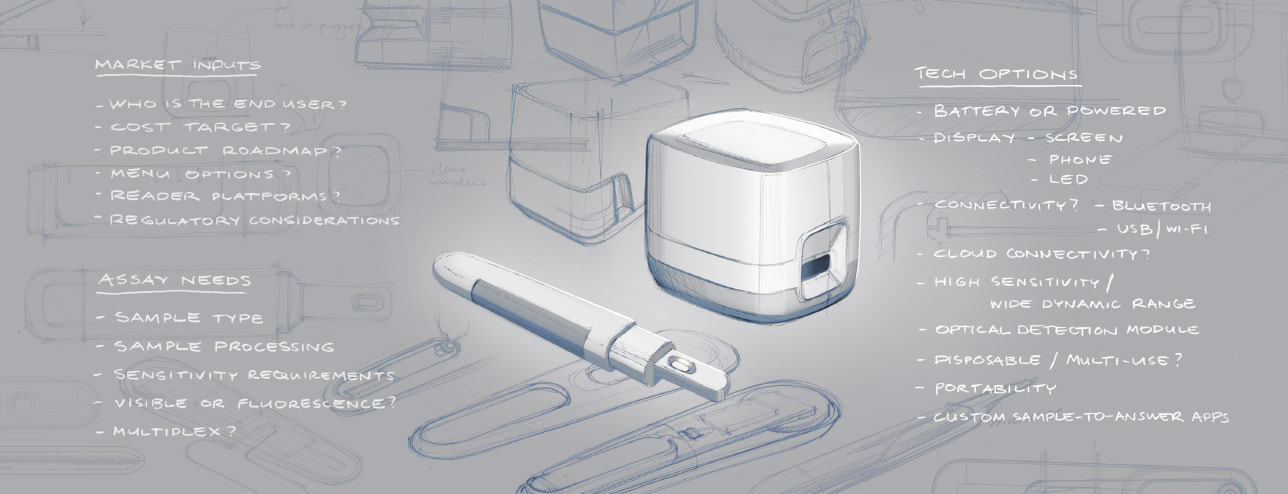
Customized Reader Solutions
Customized Reader Solutions Lumos understands that developing all the components needed for a successful POC device can be an expensive endeavor. We draw upon our extensive understanding of the POC market to develop our own POC reader platforms. With built-in flexibility, we can easily tailor the final product to meet clients' needs. We provide mixed and match features and functions to help develop the reader solution that best meets your target market requirements.